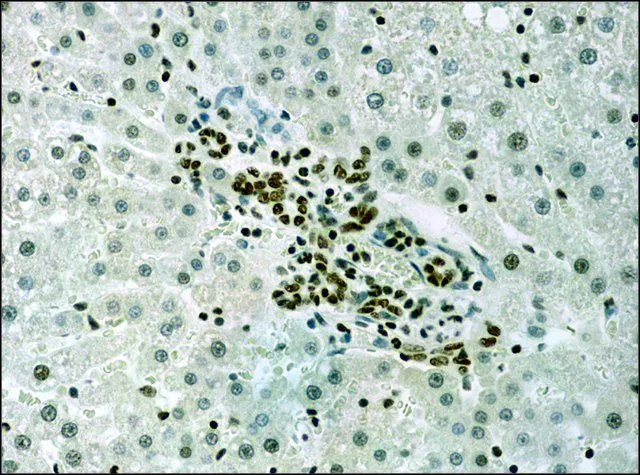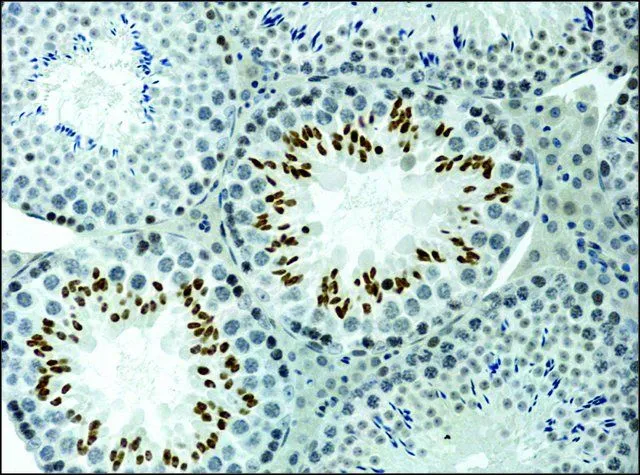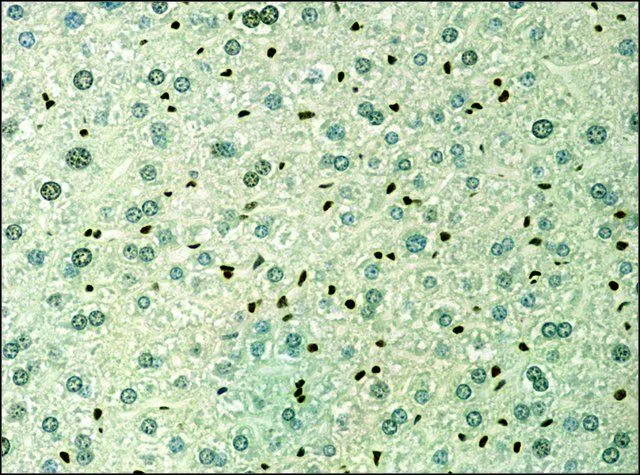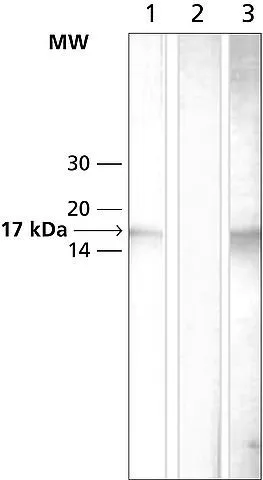
GTX12178 IHC-P Image
Histone H3K9ac (Acetyl Lys9) antibody

GTX12178
ApplicationsImmunoFluorescence, Western Blot, ImmunoCytoChemistry, ImmunoHistoChemistry, ImmunoHistoChemistry Paraffin
Product group Antibodies
ReactivityAmphibian, Bovine, C. Elegans, Chicken, Drosophila, Human, Mouse, Rat
Overview
- SupplierGeneTex
- Product NameHistone H3K9ac (Acetyl Lys9) antibody
- Delivery Days Customer10
- ApplicationsImmunoFluorescence, Western Blot, ImmunoCytoChemistry, ImmunoHistoChemistry, ImmunoHistoChemistry Paraffin
- CertificationResearch Use Only
- ClonalityPolyclonal
- Concentration2-2.5 mg/ml
- HostRabbit
- IsotypeIgG
- Modification TypeAcetylated
- Scientific DescriptionHistone proteins H3, H4, H2A, and H2B function as building blocks to package eukaryotic DNA into repeating nucleosome units that are folded in higher order chromatin fibers. The nucleosome is composed of an octamer containing a H3/H4 tetramer and two H2A/H2B dimers, surrounded by approximately 146 base pairs of DNA. A diverse and elaborate array of post-translational modifications including acetylation, phosphorylation, methylation, ubiquitination, and ADP-ribosylation occurs on the N-terminal tail domains of histones. Acetylation of lysine residues within these N-terminal domains by histone acetyl-transferases (HATs), including Gcn5p, P/CAF, p300/CBP, and TAFII250, is associated with transcriptional activation. This modification results in remodeling of the nucleosome structure into an open conformation more accessible to transcription complexes. Conversely, histone deacetylation by histone deacetylases (HDACs) is associated with transcription repression reversing the chromatin remodeling process. In most species, histone H3 is primarily acetylated at lysine 9, 14, 18, and 23. Acetylation at lysine 9 appears to have a dominant role in histone deposition and chromatin assembly in some organisms.
- ReactivityAmphibian, Bovine, C. Elegans, Chicken, Drosophila, Human, Mouse, Rat
- Storage Instruction2°C to 8°C,-20°C
- UNSPSC12352203