BioVendor’s innovative ELISA and POCT solutions reduce diagnostic uncertainty in bacterial and viral infections, with MxA setting a new standard for diagnostic innovation.
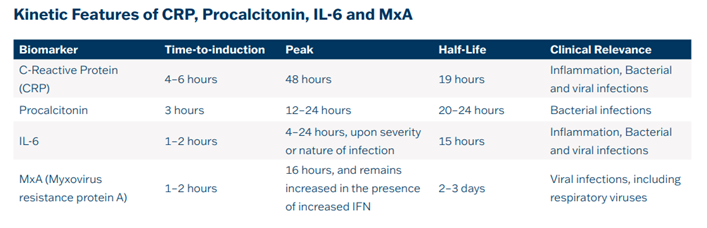
In current laboratory diagnostics, measurements of CRP, PCT, and IL-6 are routinely used to manage inflammation and infection. Diagnostic performance can be further improved by determining human MxA protein, which offers advantages due to its low baseline concentration that increases within 1–2 hours after the onset of viral infection but not bacterial infection and long half-life in circulation being approximately 2–3 days.
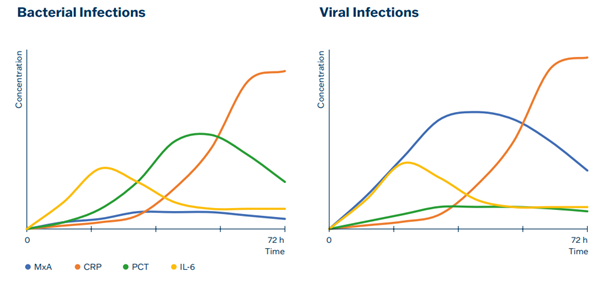
The severity of the clinical condition was accompanied by high levels of MxA, while a decrease in MxA was observed when the clinical condition improved. CRP levels showed a delayed response compared to MxA and PCT concentrations indicates the absence of bacterial coinfection.
New Insights into Clinical Diagnostics
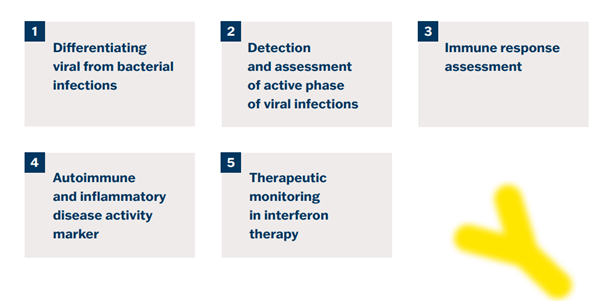
1. Differentiting Viral from Bacterial Infections
Elevated MxA levels are more specific to viral infections and help differentiate viral infections from bacterial infections. MxA is useful in clinical practice to avoid unnecessary antibiotic treatments in viral infections. MxA has been particularly studied in the context of respiratory infections, such as influenza and respiratory syncytial virus (RSV), as its levels rise significantly in these conditions.
The frequency of viral infections is 6–8 times more frequent over bacterial infections in newborns or preterm newborns, and antibiotic treatment is overused substantially. Avoid unnecessary antibiotic treatments substantially reduces risks of immunity damage or later autoimmunity disorders development.
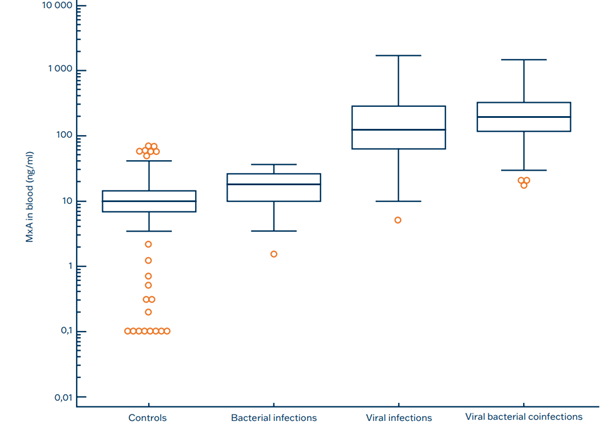
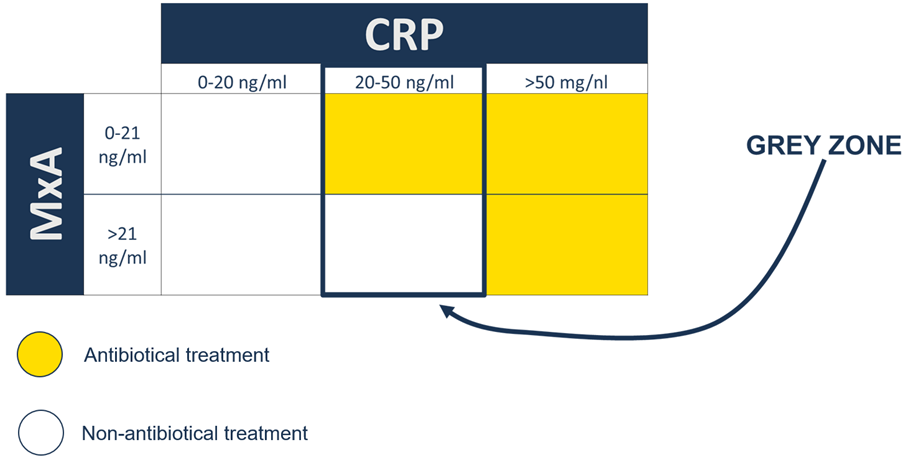
Clinical outcome from MxA measurements
- Negative predictive value (99%) = MxA in a combination with CRP – excluding individulas from antibiotic treatment
- Clinical sensitivity /CRP and clinical specifity /MxA improves diagnostic efficiency
2. Detection and Assessment of Active Phase of Viral Infections
MxA expression is significantly upregulated in peripheral blood mononuclear cells in response to type I interferons (IFN-α/β), which are typically produced in response to viral infections, or directly by viruses. Therefore, measuring MxA levels help identify viral infections, even when the specific virus is not easily detectable.
3. Immune Response Assessment
MxA serves as a marker for assessing the overall immune response capability, particularly in immunodeficiencies or after organ transplantations.
4. Autoimmune and Inflammatory Disease Activity Marker
In diseases such as systemic lupus erythematosus (SLE), which are linked to the pathogenesis of type I interferons, MxA levels reflect disease activity and can monitor disease progression or response to treatment.
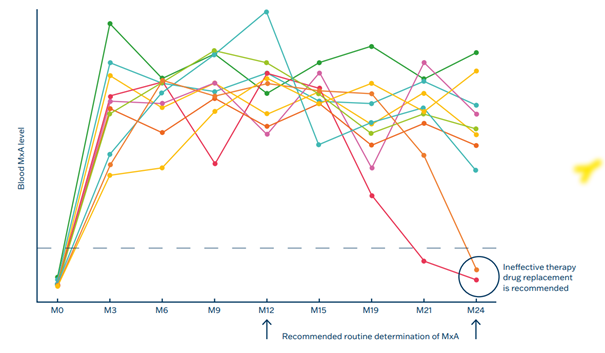
5. Therapeutic Monitoring in Interferon Therapy
In patients receiving interferon therapy, such as those with multiple sclerosis10 or certain cancers, MxA levels can be monitored to assess the treatment’s efficacy. An increase in MxA indicates a response to therapy. 2–40% percent of MS patients develop IFN-beta neutralizing antibodies (NAb) with subsequent attenuation of MxA protein induction. MxA assay can be used for assessment of raised NAb according to the recommendation from EMA (European Medicine Agency).
Who Can Benefit from MxA Assay?
- Patients – with upper respiratory tract infections;
- Hospitals – for monitoring organ transplant recipients, and preoperative and/or postoperative investigations;
- Private Medical Care Centres – with travel and tropical medicine department for screening blood-borne tropical viral-origin fevers, such as dengue fever, malaria, or yellow fever;
- Specialized Hospitals Healthcare Centres – for conditions Sjögren‘s Syndrome (applicability of MxA in stratifying patients with primary Sjögren‘s syndrome according to IFN positivity) and multiple sclerosis (monitoring the efficacy of treatment in patients with multiple sclerosis treated with interferon beta).
Human MxA ELISA
| Product Name | Catalog No. | Size | Regulatory Status |
| Human MxA ELISA | RD194349220R | 96 wells | RUO |
Human MxA POCT
Bi-VirTest for Human MxA POCT is a lateral flow assay developed for the quantitative measurement of MxA in capillary blood. It employs a pair of specific monoclonal antibodies along with a gold nanoparticle conjugate for detection. Quantitative results are obtained by reading the signal using either the Igloo or Bi-Reader® device.
| Product Name | Catalog No. | Size | Regulatory Status |
| BiVirTest | BI005-10 | 10 tests / package | CE IVD |
Igloo
Igloo Pro is an advanced portable point-of-care analyzer compatible with a wide range of lateral. It is open compatible with 90% of POCT tests from any manufacturer after successful adaptation. The instruments offers an universal connectivity to PC and mobils devices with Android or iOS, including automatic calibration, secure data managment or data export.


| Product Name | Catalog No. | Size | Regulatory Status |
| Igloo Pro | GCDD0001 | 1 piece | CE IVD |

ELISA, CLIA, BLOT, Microblot-Array | Proteins | Antibodies | IVD | PCR, LAMP, miRNA detection| IHC | endotoxin testing | POCT CE/IVD







