Click Chemistry reagents – highly selective, rapid and biocompatible labeling
Click Chemistry [1] describes pairs of functional groups that rapidly and selectively react (“click”) with each other in mild, aqueous conditions. The concept of Click Chemistry has been transformed into convenient, versatile and reliable two-step coupling procedures of two molecules A and B [1-5], that are widely used in biosciences [6-8], drug discovery [9] and material science [10].
Principle of Click Chemistry
- Activation of molecule A and B
Compatible CLICK-functional groups are introduced via CLICK Reagents - CLICK-coupling of molecule A and B
The CLICK-activated molecules A and B form a stable conjugate
Advantages of Click Chemistry
- Highly selective, low background labeling:
CLICK-functional groups are inert to naturally occurring functional groups (“bioorthogonal”) such as amines - Rapid and quantitative labeling
- Allows non-radioactive analysis of enzymatic activities both in vitro and in vivo:
Small-sized CLICK-functional groups possess excellent substrate properties
Especially biomolecule labeling requires reaction procedures that can be performed under physiological conditions (neutral pH, aqueous solution, ambient temperature) with low reactant concentrations to ensure non-toxic, low background labeling at reasonable time scales while still preserving biological function. Among the plethora of possible reactions only a few generally fit the necessary reactivity, selectivity and biocompatibility criteria (Fig. 1):
- Cu(I)- catalyzed Azide – Alkyne Click Chemistry reaction (CuAAC)
- Strain-promoted Azide – Alkyne Click Chemistry reaction (SPAAC)
- Tetrazine – trans-Cyclooctene Ligation
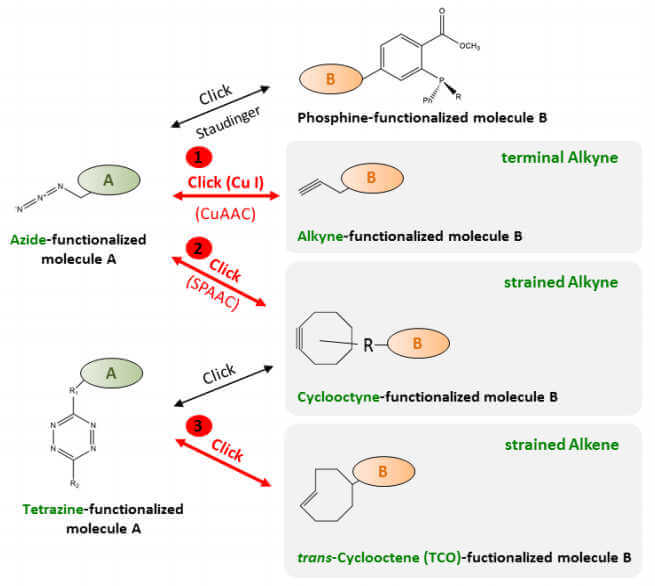
Click Chemistry reactions can be categorized into two separate groups: Copper (Cu(I))-catalyzed and Copperfree. The Cu(I)- catalyzed Azide – Alkyne Click Chemistry reaction (CuAAC) (1.) relies on the presence of Cu(I) ions whereas the Copper-free strain-promoted Azide – Alkyne Click Chemistry reaction (SPAAC) (2.) and Tetrazine – trans-Cyclooctene (TCO) Ligation (3.) efficiently proceed without metal catalysis. The well-known Copper-free Azide-Phosphine reaction (Staudinger Ligation) is hampered by the instability of phosphines and slow reaction kinetics. Recent focus therefore shifted towards strain-promoted reactions with cyclooctynes and Tetrazine – TCO Ligation, respectively.
Jena Bioscience selected the best performing CLICK reactions in terms of selectivity, reactivity, biocompatibility and stability!
1. Cu(I)-catalyzed Azide-Alkyne Click Chemistry (CuAAC) reaction
Clearly the most prominent example of click chemistry is the Cu(I)-catalyzed Azide-Alkyne Click Chemistry (CuAAC) reaction [1]. An Azide-functionalized molecule A reacts with a terminal Alkyne-functionalized molecule B thereby forming a stable conjugate A-B via a Triazole moiety (Fig. 2).
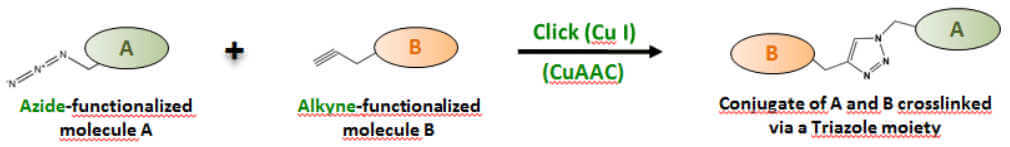
Since terminal Alkynes are fairly unreactive towards Azides, the efficiency of a CuAAC reaction strongly depends on the presence of a metal catalyst such as copper (Cu) in the +1 oxidation state (Cu(I)). Different copper sources and reduction reagents are available however, the Cu(II) salt CuSO4 as copper source in combination with ascorbate as a reduction reagent has been recommended for most biomolecule labeling applications [11,12].
The use of CuAAC reactions in live cells is hampered by the toxicity of Cu(I) ions. This problem has been partially overcome by the use of Cu(I) chelating ligands such as THPTA that serve a dual purpose: 1) Acceleration of the CuAAC reaction by maintaining the Cu(I) oxidation state and 2) Protection of the biomolecule from oxidative damage.
Presolski et. al. [11] and Hong et. al. [12] provide a general protocol for CuAAC reactions that may be used as a starting point for the set up and optimization of individual assays.
Features
- Small-sized azides and alkynes possess excellent substrate propertie
- Optimization of assay conditions required (type & concentration of Copper source, reduction reagent and Copper ligand)
- Suitable if potential copper toxicity does not matter (not recommended for in vivo or live cell labeling)
- Slowest reaction speed compared to 2. and 3.
2. Strain-promoted Azide-Alkyne Click Chemistry (SPAAC) reaction
The requirement of a cytotoxic copper catalyst often limits the usage of CuAAC reactions (see 2.) A Copper free and thus non-toxic labeling method of Azides is the Strain-Promoted Azide – Alkyne Click Chemistry (SPAAC) reaction [3]. SPAAC reactions rely on the use of strained cyclooctynes that possess a remarkably decreased activation energy in contrast to terminal Alkynes and thus do not require an exogenous catalyst [13].
A number of structurally varied cyclooctyne derivatives (e.g. DIFO, BCN, DIBAC, DIBO, ADIBO) have been developed that strongly differ in terms of reaction kinetics and hydrophility. Jena Bioscience’s SPAAC conjugation chemistry is based on the reaction of Azadibenzylcyclooctyne (ADIBO = DBCO = DIBAC) (Fig. 3).
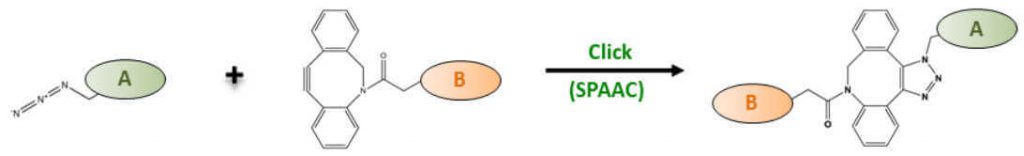
Azadibenzocyclooctyne (ADIBO=DBCO)-based reagents combine high reactivity with sufficient hydrophility [14,15] and thus allow low background labeling of Azide-functionalized molecules [16] with even greater efficiency than CuAAC reactions. Azide-DBCO reactions are furthermore highly selective and therefore ideally suited for dual labeling approaches with Tetrazine – trans-Cyclooctene Ligation (see 3.) [17].
Features
- Faster detection of small-sized Azides compared to CuAAC reactions (see 2.)
- Copper free and thus non-toxic
- No catalyst or accessory reagents and thus no extensive optimization of assay conditions required
- Suitable for dual-labeling approaches in combination with Tetrazine – trans-Cyclooctene Ligation
3. Tetrazine-trans-Cyclooctene Ligation
The Tetrazine – trans-Cyclooctene Ligation constitutes a non-toxic biomolecule labeling method of unparalleled speed that is ideally suited for in vivo cell labeling and low concentration applications. A Tetrazine-functionalized molecule A reacts with a trans-Cyclooctene (TCO)-functionalized molecule B thereby forming a stable conjugate A-B via a Dihydropyrazine moiety (Fig. 4).
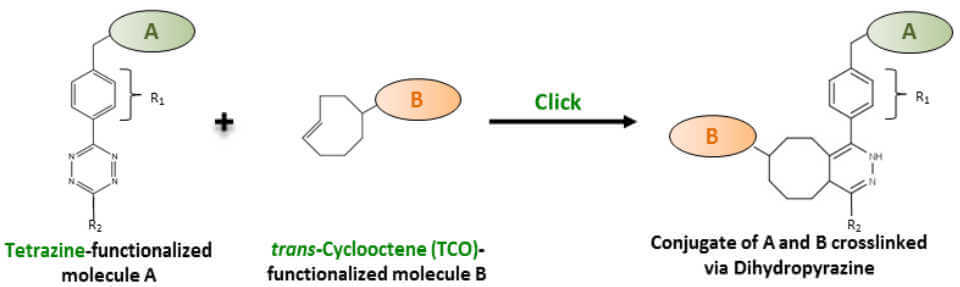
A number of structurally varied strained alkene and tetrazine derivatives have been developed that strongly differ in terms of reaction kinetics and stability. TCO has been selected (as strained alkene) since it possesses the highest reactivity towards tetrazine [18,19].
The reactivity of the tetrazine derivatives towards TCO is determined by the substituents in the 3 position (Fig. 4, R1) and 6 position (Fig. 4, R2). Two Tetrazine versions with different reactivities and stability characteristics have been selected that meet specific application requirements. Tetrazine (R1=phenyl, R2=H) reagents are the ideal choice if a rapid reaction kinetic is the key aspect, whereas 6-Methyl-Tetrazine (R1=phenyl, R2=CH3) reagents are ideally suited if an improved chemical stability is required [18].
Features
- High-speed CLICK reaction that is ideally suited for in vivo cell labeling & low concentration applications
- Copper free and thus non-toxic
- No catalyst or accessory reagents and thus no extensive optimization of assay conditions required
- Suitable for dual-labeling approaches in combination with the strain-promoted Azide – DBCO reaction17
Overview of available CLICK reagents
| Azides | Alkyne | Dibenzocyclooctyne | Tetrazine | Trans-Cyclooctene | Vinyl | Methylcyclopropene | Norbornene | |
| Fluorescent dyes | √ | √ | √ | √ | ||||
| (Desthio)Biotion | √ | √ | √ | √ | ||||
| FLAG tag | √ | √ | √ | |||||
| Bifunctional reagents | √ | √ | √ | √ | √ | |||
| Trifunctional reagents | √ | |||||||
| PEGylation reagents | √ | |||||||
| Nucleotides | √ | √ | √ | √ | √ | |||
| Nucleosides | √ | √ | √ | √ | ||||
| Nucleobases | √ | |||||||
| Amino acids | √ | √ | ||||||
| Monosacharides | √ | √ | √ | |||||
| Agarose & magnetic beads | √ | √ | √ | √ | √ |
If you did not find the products that you are looking for, please contact our techsupport.
Selected references
- Kolb HC, Finn MG, Sharpless KB. Click chemistry: diverse chemical function from a few good reactions. Angew Chem Int Ed Engl. 2001;40(11):2004-2021.
- Sletten EM, Bertozzi CR. Bioorthogonal chemistry: fishing for selectivity in a sea of functionality. Angew Chem Int Ed Engl. 2009;48(38):6974-6998.
- Jewett JC, Bertozzi CR. Cu-free click cycloaddition reactions in chemical biology. Chem Soc Rev. 2010;39(4):1272-1279.
- Best MD. Click chemistry and bioorthogonal reactions: unprecedented selectivity in the labeling of biological molecules. Biochemistry. 2009;48(28):6571-6584.
- Lallana E, Riguera R, Fernandez-Megia E. Reliable and efficient procedures for the conjugation of biomolecules through Huisgen azide-alkyne cycloadditions. Angew Chem Int Ed Engl. 2011;50(38):8794-8804.
- Grammel M, Hang HC. Chemical reporters for biological discovery. Nat Chem Biol. 2013;9(8):475-484.
- Xie R, Hong S, Chen X. Cell-selective metabolic labeling of biomolecules with bioorthogonal functionalities. Curr Opin Chem Biol. 2013;17(5):747-752.
- Su Y, Ge J, Zhu B, Zheng Y-G, Zhu Q, Yao SQ. Target identification of biologically active small molecules via in situ methods. Curr Opin Chem Biol. 2013;17(5):768-775.
- Zeng D, Zeglis BM, Lewis JS, Anderson CJ. The growing impact of bioorthogonal click chemistry on the development of radiopharmaceuticals. J Nucl Med. 2013;54(6):829-832.
- Evans RA. The rise of azide—alkyne 1,3-dipolar “click” cycloaddition and its application to polymer science and surface modification. ChemInform. 2007;38(40).
- Presolski SI, Hong VP, Finn MG. Copper-catalyzed azide–alkyne click chemistry for bioconjugation. Curr Protoc Chem Biol. 2011;3(4):153-162.
- Hong V, Presolski SI, Ma C, Finn MG. Analysis and optimization of copper-catalyzed azide-alkyne cycloaddition for bioconjugation. Angew Chem Int Ed Engl. 2009;48(52):9879-9883.
- Ess DH, Jones GO, Houk KN. Transition states of strain-promoted metal-free click chemistry: 1,3-dipolar cycloadditions of phenyl azide and cyclooctynes. Org Lett. 2008;10(8):1633-1636.
- Debets MF, van Berkel SS, Schoffelen S, Rutjes FPJT, van Hest JCM, van Delft FL. Aza-dibenzocyclooctynes for fast and efficient enzyme PEGylation via copper-free (3+2) cycloaddition. Chem Commun (Camb). 2010;46(1):97-99.
- Kuzmin A, Poloukhtine A, Wolfert MA, Popik VV. Surface functionalization using catalyst-free azide−alkyne cycloaddition. Bioconjugate Chem. 2010;21(11):2076-2085.
- Yao JZ, Uttamapinant C, Poloukhtine A, et al. Fluorophore targeting to cellular proteins via enzyme-mediated azide ligation and strain-promoted cycloaddition. J Am Chem Soc. 2012;134(8):3720-3728.
- Liang Y, Mackey JL, Lopez SA, Liu F, Houk KN. Control and design of mutual orthogonality in bioorthogonal cycloadditions. J Am Chem Soc. 2012;134(43):17904-17907.
- Selvaraj R, Fox JM. Trans-cyclooctene–a stable, voracious dienophile for bioorthogonal labeling. Curr Opin Chem Biol. 2013;17(5):753-760.
- Karver MR, Weissleder R, Hilderbrand SA. Synthesis and evaluation of a series of 1,2,4,5-tetrazines for bioorthogonal conjugation. Bioconjug Chem. 2011;22(11):2263-2270.
- Sečkutė J, Devaraj NK. Expanding room for tetrazine ligations in the in vivo chemistry toolbox. Curr Opin Chem Biol. 2013;17(5):761-767.

PCR | Molecular Biology Reagents | Proteins | LEXSY | Nucleotides & Nucleosides | Crystallography | Cryo-EM | RNA | Labeling







