Cygnus Technologies: When it comes to developing your internal analytical programs, it is important to understand the complexity of Host Cell Protein (HCP) analysis and the risks associated with insufficient HCP characterization.
With a track record of over 25 years in value-added analytics for HCPs, biopharmaceutical industry relies on Cygnus Technologies to develop robust, broadly reactive, and qualified HCP immunoassays for many cell expression platforms. From early process development utilizing generic HCP ELISA to long-term and uninterrupted supply of process-specific HCP ELISA critical to QC release testing of the approved drug, Cygnus Technologies will support your success at every stage.
- Process-specific HCP Antibody and Assay Development
- HCP Antibody Coverage Analysis by AAE™ and AAE-MS™ Methods
- HCP Identification and Quantification in Final Drug Substances
- Assay Qualification
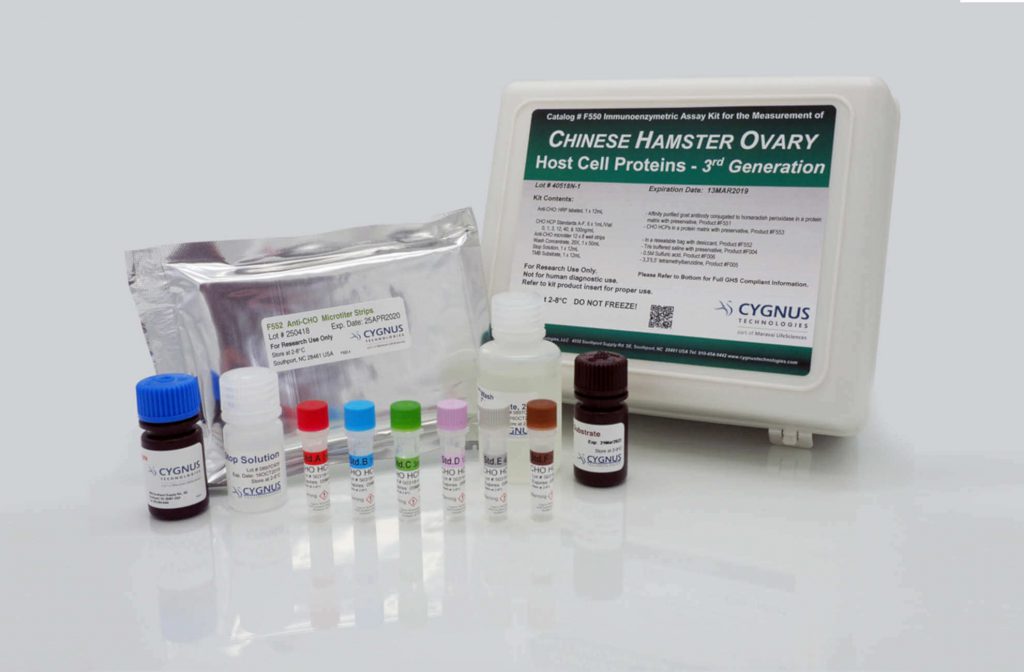
A robust and broadly reactive host cell protein (HCP) Enzyme-Linked Immunosorbent Assay (ELISA) is a critical tool for monitoring purification process consistency as well as final drug substance purity and is the gold standard method for product monitoring and release testing for HCPs. HCP ELISA can measure ng/ml levels of HCP in the presence of mg/ml levels of product protein, does not require a special expertise to run, and is easy to transfer across an organization from process development to manufacturing quality control groups.
“Immunoassay methods rely on antibodies that recognize, as broadly as possible, the population of HCPs entering the downstream purification process; therefore, the sandwich immunoassay, designed with polyclonal antibodies, is the workhorse of HCP monitoring and quantitation. This assay format offers a combination of high sensitivity, specificity, throughput, automation potential, rapid turnaround, quantitative results, and low cost per assay that is unmatched by any other currently available assay technology.” – USP Chapter 1132, 3. HCP Immunoassay Methods
At the same time, HCP ELISA requires a high level of expertise to develop a broadly reactive HCP antibody and then turn it into an accurate, sensitive, specific and robust assay. In addition, HCP ELISA does not give information regarding the individual HCPs present in the final drug substance, which HCPs are being recognized, or if the assay is broadly reactive.
Regulatory agencies around the world have put measures in place to ensure the HCP ELISA used by a sponsor is fit for the purpose of monitoring purification process consistency and product lot release. It is advised to employ orthogonal methods [2] such as Antibody Affinity Extraction (AAE™) immunoaffinity chromatography combined with 2D-PAGE and/or Mass Spectrometry (MS) methods to demonstrate antibody coverage to individual HCPs to support that the HCP ELISA is fit-for-purpose. Equally important is to qualify the assay for dilution linearity, accuracy and precision.
Cygnus offers generic HCP ELISA Kits for 21 different cell lines, advanced orthogonal antibody coverage analysis services, HCP identification in process samples and drug substances by AAE-MS™, generic assay qualification services and expertise in process-specific antibody and assay development services.
Cygnus’ reputation for quality is recognized by the industry and global regulatory agencies, with several generic HCP ELISA Kits supporting marketed biologics. Cygnus’ proprietary technology has also been utilized to develop over 100 process-specific antibodies and immunoassays for many global biopharmaceutical companies.
Bio-Connect Life Sciences supplies all their products in the Benelux. If you have any questions, please contact the helpdesk.

Host Cell DNA detection | Viral clearance | MS | Host Cell Protein (HCP) | ELISA | Biotherapeutics | Antibodies | WB | Immunoassay Reagents