All InVivo grade antibodies feature greater than 95% purity, ultra-low endotoxin levels, and are preservative, stabilizer, and carrier protein-free.
The Bio X Cell InVivoPlus™ line of antibodies are of a grade and quality that will meet or exceed the strict demands and standards of many in vivo and in vitro experiments. They are extra low in endotoxin (≤0.001 EU/µg), contain ≤ 5% protein aggregates and have been validated by Flow or ELISA. The InVivoPlus™ antibodies are also free of murine pathogens, azide and carrier proteins, and are highly pure. The InVivoPlus™ line of antibodies are perfectly suited for highly sensitive assays and in vivo studies with live cells or whole animals on a variety of functional assays.
Selection of InVivoPlus™ antibodies
What is the difference between InVivoMab and InVivoPlus?
Bio X Cell offers InVivoPlus™ versions of the most popular InVivoMAb™ products. The InVivoPlus™ versions are structurally and functionally identical to the InVivoMAb™ versions. However, the difference lies in a superior level of quality that will meet or exceed the strict demands and rigorous standards required for in vivo work at any research organization.
| Comparison | InVivoMAb™ | InVivoPlus™ |
|---|---|---|
| Binding Validation* Determined by immunoblot, flow cytometry, or ELISA | No | Yes |
| Murine Pathogen Screening† Determined by ultrasensitive real-time PCR | No | Yes |
| Antibody Aggregation Screening Determined by Dynamic Light Scattering | No | Yes |
| Endotoxin Determined by LAL gel clotting assay | <2EU/mg | <1EU/mg |
| Purity Determined by SDS-PAGE | >95% | >95% |
| Preservative, stabilizer and carrier protein free | Yes | Yes |
| Formulated for in vivo use | Yes | Yes |
| Catalog number format | Begins with BE | Begins with BP |

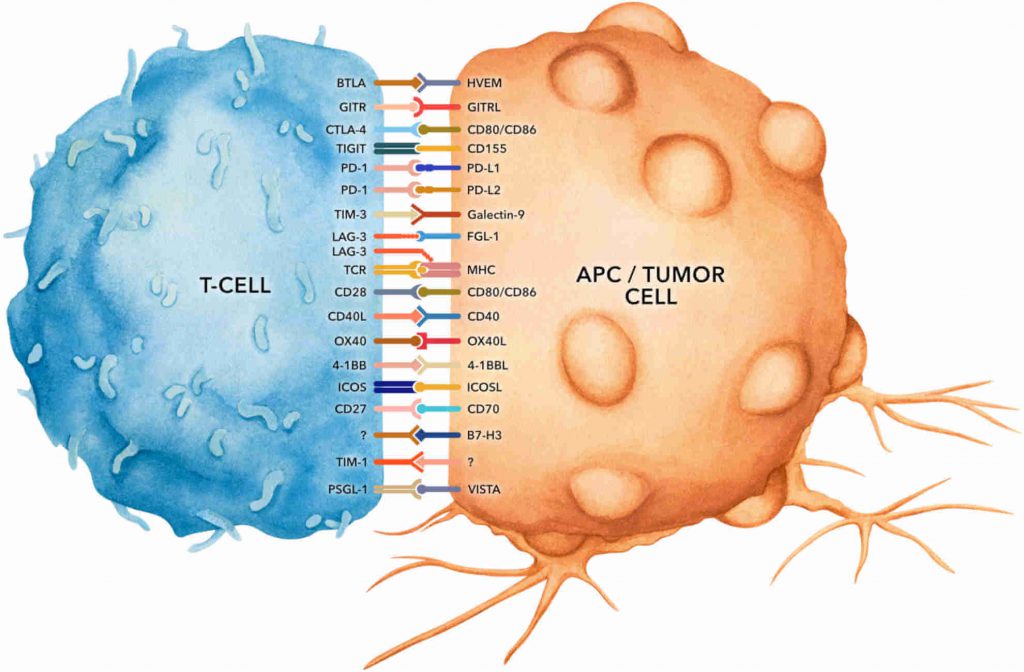
In Vivo Monoclonal Antibodies (Mabs) | Isotype Controls | InVivoSIM™ Biosimilar Antibodies | RecombiMAb™ | Functional Bispecific Solutions | PD-1 | TIGIT, TIM-3, LAG-3 | Reagents